Smart life sciences

Whether high-resolution imaging, gentle and precise laser treatment, digital screening, or individualized medical technology, we are working on smart solutions for future life sciences and biomedical issues. These include systems for high-throughput screening and novel light sources for high-resolution imaging directly in the operating room. We are also involved in the manufacture of patient-specific implants and the functionalization of implant surfaces. With our research, we want to enable and establish individual, safe, and novel treatment methods.
Your contact person
Dr. rer. nat. Tammo Ripken
Overview of smart life sciences
Laser medicine
In the future, smart laser medicine will enable intelligent controls that optimally support the physician. The goals of our technology developments are to shorten surgery and treatment times. We develop innovative laser- or light-based treatment methods and transfer them into practice. This is because laser medicine enables the extremely gentle treatment of tissue and thus minimally invasive individual therapies for patients. In addition to practical research and development, we advise and assist in approving new procedures, more on this in our Regulatory Affairs section.

Lasers are now used as a standard for operations on the eye, one example being the fs-LASIK procedure we co-developed. To be able to use the laser for other diseases of the eye, we are developing new methods or transferring established treatments to other target areas in the eye. For example, we are working on treating floaters (or: mouches volantes), presbyopia or keratoconus. To do this, we use ultrashort pulse lasers, which can be used to treat tissues very gently and precisely. We also use optical imaging, such as optical coherence tomography, for the best possible support in diagnosis, navigation, and therapy control. In addition to process and system development, we also work on the biomechanics of tissues in the eye.

Lasers already make it possible to remove skin or tissue in a targeted and highly gentle manner in many areas of surgery. To expand the use of lasers even further, we are researching hard tissue ablation of bone, for example. In this way, we want to enable the painless and straightforward replacement of implants. We are using optical coherence tomography (OCT) to allow surgeons in the future to monitor interventions in the best possible way. With OCT, the uppermost tissue layers can be imaged reliably and in real-time.
Analysis methods and imaging
In the field of imaging and analytical procedures, we work with and on various (optical) methods and techniques and even combine these methods. This makes it possible to image, analyze and correlate samples across different scales. Not only can we make statements about individual cells and the cell association, but we can also show their relationship to surrounding tissues. In addition, we can use OCT to examine tissue in motion. Our focus is also on integrating imaging into (existing) workflows. We also have a scanning electron microscope (SEM) and a nanoscale computer tomograph (nano-CT) at our disposal.
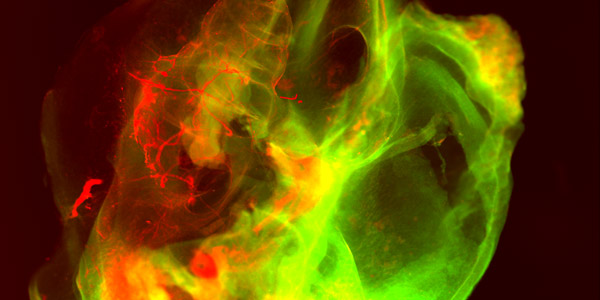
With optical tomography, we can measure mesoscale biological or technical samples and even visualize implants in tissues. We are improving our proprietary development Scanning Laser Optical Tomography (SLOT) and the light-sheet microscopy, horizontal 2-photon microscopy, and Optical Coherence Tomography (OCT) to customize them for specific problems and develop them into commercial systems. Furthermore, we optimize image processing, develop intelligent evaluation algorithms and correlative displays. The aim is, for example, to enable real-time evaluation of image data to shorten operations or achieve more precise conclusions in the life sciences through big data analyses.

We are working on correlating imaging methods to obtain the most accurate and cross-scale information from samples. For this purpose, we combine the tomographic method SLOT with fluorescence spectroscopy to record fluorescence spectra of 3D samples. Hereby we add the complete spatial information to classical microscopy methods and thus provide new tools for cell biology research. For example, this is helpful fot studying 3D cell constructs that are increasingly replacing two-dimensional targets. For this, we are developing our own smart reconstruction algorithms. We are also using Raman spectroscopy in innovative optical systems. Therefore, we are opening up new application possibilities that previously failed due to the long acquisition times through this methodological advancement.

In addition to a high resolution, laser microscopy methods enable the visualization of small three-dimensional volumes - in the best case even marker-free. We are engaged in further developing the methodology of these procedures and adapt them to new requirements in research and development. In particular, we are concerned with smart post-processing of the data and developing software for automated quantification, among other things. This should make it possible to evaluate questions with many recordings easily and quickly and thus apply Big Data approaches. In addition, we also take care of a particularly user-friendly handling of the samples both in the preparation and in the imaging process itself.

We work on novel lasers and laser systems to open up new imaging applications through more compact, cost-effective, and innovative light sources. For example, we work on ultrashort pulse sources with multiple beam outputs and rapidly tunable wavelengths from the visible to the infrared range. With these, we want to enable multimodal imaging systems. These would analyze a sample within one device using different imaging techniques. This would enable faster and more cost-effective examinations of cells and tissues - in perspective, even during an operation.
Biophotonics
In the field of biophotonics, we manipulate and control materials and cells with precision. The use of light in biology opens up new possibilities in research and the treatment of diseases. Digital processes and automation allow high throughput with high reproducibility. We develop smart methods and protocols and work on developing entire systems for application in research and industry.
We use light to precisely manipulate biomaterials and substances that come into contact with cells and tissues. We create scaffolds for tissue engineering or use hydrogels to implant them as biocompatible light guides or sensors with laser-based processes. Laser irradiation can be used to locally modify material properties. We use this principle to place cells selectively and specifically on surfaces and thus enable more detailed investigations, for example for drug screening.

Optogenetics offers a wide range of possibilities to control cells and their behavior by light. We are working on optical switches and the associated processes to control them - also in high-throughput applications. We are also involved in product development for the straightforward use of optogenetic protocols. For this purpose, we compile and evaluate kits.

The light-based manipulation of cells is very gentle and highly efficient. On this basis, we develop new applications up to automated prototypes for high-throughputs. We accompany the implementation together with partners up to market maturity. Our GNOME system, for example, enables automated and precise gold nanoparticle-assisted laser cell transfection in high-throughput. In addition to vitaly-protective manipulation, the targeted inactivation of bacterial cells is also being researched. Here, for example, light-based methods are used to keep implanted or implantable materials germ-free.

We develop high-throughput procedures for research and development and work on the automation of standard biological procedures and evaluations. In doing so, we deal with process control and integrating software, processes, and systems into existing workflows and devices. We also deal with the stimulation of cells by light. Different mechanisms such as direct (nerve) cell stimulation or stimulation by mechanical stimuli using optoacoustics are used depending on the application.
Medical technology
Laser material processing opens up far-reaching possibilities for tomorrow's medical technology. We are working on processes for 3D printing, studying the effects of laser radiation on materials for medical technology products, and developing structuring that gives products such as implants new properties. In doing so, we often rely on ultrashort pulse lasers, as they enable very gentle and flexible processing.

Additive manufacturing technology in the powder bed can be used to produce implants that are precisely tailored to the needs of patients. We deal with the production of stents as well as osteosynthesis implants, endoprosthetics, or reconstructive bone implants. We process special materials, such as the shape memory alloy nickel-titanium or bioresorbable magnesium alloys, for smart implants of the future. We are researching additive processes to reduce manufacturing costs to make individual medicine affordable for these and other materials.

We research materials for the medical technology sector and test them for 3D printing and surface processing. The requirements for materials and products in the medical industry are extremely high: Biocompatible materials must not change their safety through processing into products, and processing must not influence their favorable properties. To this end, we adapt processes, systems, and equipment to these requirements and develop them further. We work on metals, polymers, and glass and continue to develop powder bed, buildup welding, and liquid-based processes to make optimal medical products possible.

Patient-specific and functionalized aids can alleviate suffering or help patients recover more quickly. In the future, additive manufacturing processes should make it possible to implement individualized fabrications rapidly and cost-effectively. Especially for the orthopedic field, there is a lot of potential to offer relief for joints, increase mobility, or restore it. We are working on 3D-printed aids with integrated functions. For example, mounts for sensors or channels can also be directly integrated into the aids.

Surface structuring allows implants, orthoses, or other relevant surfaces to be equipped with new functions. The process can be used to specifically promote or inhibit the growth of cells or bacteria on surfaces or to specify the growth direction. This actively stimulates the colonization of implants or prevents overgrowth by unwanted tissue, as is sometimes the case with not only heart valves. We are also working to endow various materials with hydrophobic or lipophilic properties to make them easier to clean. We are developing processes that functionalize surfaces by changing their topography or chemical properties.
Regulatory Affairs
The approval of medical devices and in-vitro diagnostics must take into account the so-called "regulatory affairs", a series of normative processes. The basis for placing medical devices and in-vitro diagnostics on the market in Europe and Germany is formed by the European Medical Device Regulation (MDR) as well as the EU In-vitro Diagnostics Regulation (IVDR) and its national implementation - the Medizinprodukterecht-Durchführungsgesetz (MPDG). We carry these out for our developments as a matter of course and advise and support external parties in the application process.

We use our expertise from research and our own approval processes to support the marketing of new medical products. We advise on regulatory issues and help with document development. We take into account the regulations currently in force at the national and EU level. These are our topics:
- Consulting and support during the conformity assessment procedure up to CE marking
- Delimitation and classification of medical devices
- Carrying out research into standards and directives
- Preparation of technical documentation according to risk classification

The approval of treatment methods and medical devices is a complex process. The focus is on proving the safety and performance of a medical device, which must be based on scientifically sound clinical data. Increasingly, data from clinical investigations are required. Our trained specialists accompany clinical investigations, whether in the context of a medical device approval or feasibility study (other clinical investigations), and pass on our experience with our own developments. We answer regulatory questions and support the planning and execution of clinical trials and the evaluation of clinical data adapted to the corresponding study categorization.

